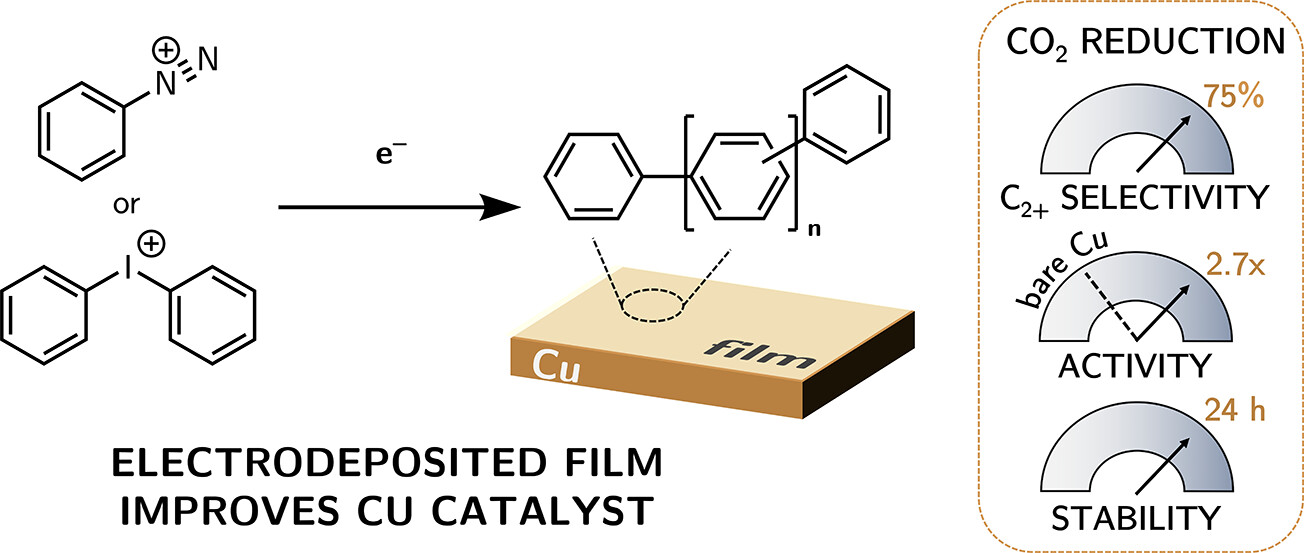
Abtract: Electrochemical reduction of CO₂ (CO₂R) by Cu metal holds promise to convert CO₂ to valuable C2+ chemicals at scale using electricity and water but suffers from poor selectivity. Coating of metal electrodes with small organic molecules or polymers has been shown to effectively enhance catalytic performance but remains underexplored. Herein, facile modification of Cu surfaces by a polyaromatic layer was found to boost both selectivity and activity toward C2+ products. Using phenyldiazonium or diphenyliodonium salts to graft an organic layer onto the surface of Cu foil electrodes resulted in up to 75% Faradaic efficiency (FE) for C2+ at neutral pH. Modified electrodes have electrochemical active surface areas and proton diffusion coefficients similar to those of bare Cu. High CO₂R performance was maintained in a gas diffusion electrode, with a pH ≈ 1 electrolyte (1 M H3PO4, 1 M KCl; −100 mA/cm2), producing 65% FE for C2+ over 5 h, with no delamination.