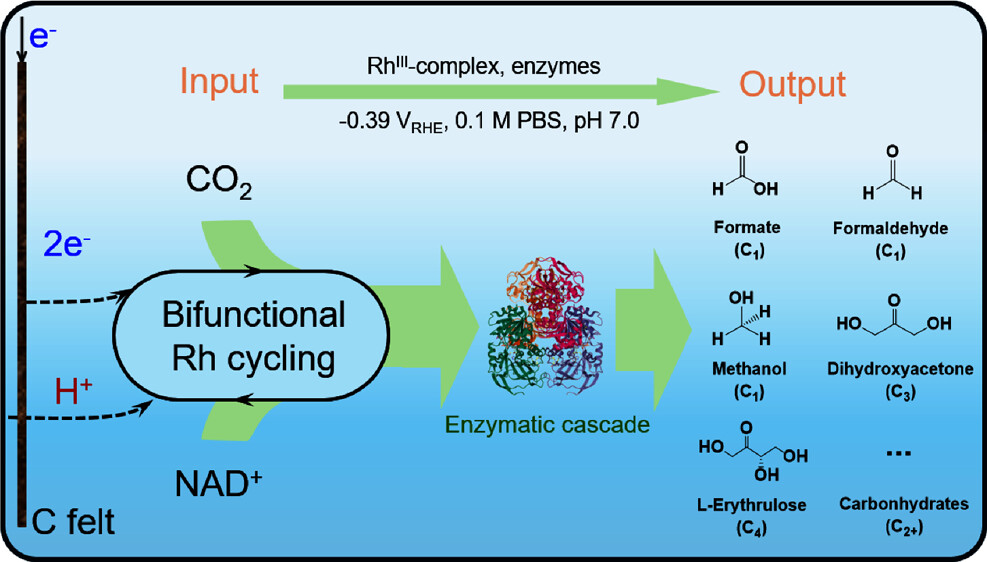
Abstract: Bioelectrochemical synthesis is emerging as an ecofriendly method for CO2 fixation, typically using electrochemically regenerated NAD(P)H to supply reducing equivalents for formate dehydrogenase (FDH) to convert CO2 to formate. However, the efficiency of this process is hindered by unfavorable thermodynamic conditions. In this study, we developed a one-pot bioelectrochemical system featuring a rhodium-based catalyst [Cp*Rh(bpy)Cl]2+ (RhIII-complex or [RhIII-H2O]2+), which works cooperatively with enzymatic cascades involving acetyl-CoA synthase (ACS), acetaldehyde dehydrogenase (ACDH), alcohol dehydrogenase (ADH), formolase (FLS), and d-fructose-6-phosphate aldolase mutant FSAA129S to convert CO2 into various C2+ chemicals. The bifunctional RhIII-complex concurrently catalyzes gas-phase reduction of CO2 to formate at 15.8 mM/h and NADH regeneration at 0.24 mM/min. Formate production is 83.2 times faster than using one of the best aerobic FDHs from Clostridium ljungdahlii (ClFDH), resulting in a 3.6-fold increase in methanol production rate (0.43 mM/h) compared to the tandem enzymatic system (0.12 mM/h). Additionally, the bifunctional RhIII-complex cooperates with enzymatic cascades to produce dihydroxyacetone (C3) and L-erythrulose (C4) with yields of 2.63 and 1.93 mM, respectively. This study highlights the synergy between electrochemical and enzymatic catalysis, providing an alternative for electroenzymatic CO2 reduction to produce value-added C2+ compounds with enhanced productivity.