Madeline H. Hicks†, Weixuan Nie†, Annette E. Boehme, Harry A. Atwater*, Theodor Agapie* and Jonas C. Peters*, J. Am. Chem. Soc. 2024, 146, 36, 25282–25289. (†: Co-First Author)
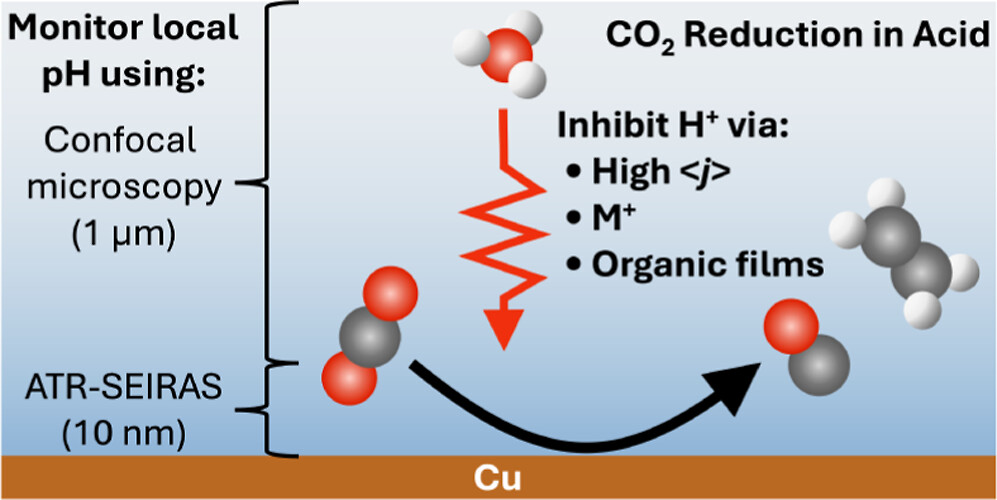
Abstract: Inspired by recent advances in electrochemical CO₂ reduction (CO₂R) under acidic conditions, herein we leverage in situ spectroscopy to inform the optimization of CO₂R at low pH. Using attenuated total reflection surface-enhanced infrared absorption spectroscopy (ATR-SEIRAS) and fluorescent confocal laser scanning microscopy, we investigate the role that alkali cations (M+) play on electrochemical CO₂R. This study hence provides important information related to the local electrode surface pH under bulk acidic conditions for CO₂R, both in the presence and absence of an organic film layer, at variable [M+]. We show that in an acidic electrolyte, an appropriate current density can enable CO₂R in the absence of metal cations. In situ local pH measurements suggest the local [H+] must be sufficiently depleted to promote H2O reduction as the competing reaction with CO₂R. Incrementally incorporating [K+] leads to increases in the local pH that promotes CO₂R but only at proton consumption rates sufficient to drive the pH up dramatically. Stark tuning measurements and analysis of surface water structure reveal no change in the electric field with [M+] and a desorption of interfacial water, indicating that improved CO₂R performance is driven by suppression of H+ mass transport and modification of the interfacial solvation structure. In situ pH measurements confirm increasing local pH, and therefore decreased local [CO₂], with [M+], motivating alternate means of modulating proton transport. We show that an organic film formed via in situ electrodeposition of an organic additive provides a means to achieve selective CO₂R (FECO₂R ∼ 65%) over hydrogen evolution reaction in the presence of strong acid (pH=1) and low cation concentrations (≤0.1 M) at both low and high current densities.